The human body is a complex organisation made up in about equal numbers of human cells, and bacterial, fungal and protozoan cells. In addition, it is considered likely that every one of us is infected by some kind of virus, but most never present any symptoms of the infection. However, we know that a limited number of these viruses, bacteria, prions and fungi can have a profound effect on our bodies (and our work schedules – I’ve got three young children), because of the diseases they cause.
How does this relate to LAMP (loop-mediated isothermal amplification)? Well, the issue that these pathogens pose to modern medicine is that current diagnostic technologies are lacking in their ability to detect specific infections, and are not widely available. As a result, patients are being over-prescribed antibiotics, which is causing an increasing level of antimicrobial resistance in the human population. This in turn requires diagnostic methods to quickly identify patients infected with antibiotic-resistant strains of bacteria. On top of that, there are specific instances where the correct identification of pathogens is time-critical, e.g bacterial meningitis.
LAMP has the potential to recognise specific kinds of human pathogens, which would in theory lead to the development of more focussed treatments for infections. Furthermore, since LAMP can work in a low-tech environment, using portable real-time instruments, it can serve as a true point-of-need method.
Current research into LAMP assays for human pathogens
 After the invention of the groundbreaking DNA amplification technique, LAMP back in 2000, there has been a boom in research into the development of applications of this method. What’s special about LAMP is that it is a low-cost and simple method which means it can be easily transported to areas which may lack resources, such as developing countries that may also have high incidences of certain pathogens. Scientists have worked on developing methods for detecting a number of human pathogens, with the aim of innovating more effective diagnosis and treatment of related infections and diseases.
After the invention of the groundbreaking DNA amplification technique, LAMP back in 2000, there has been a boom in research into the development of applications of this method. What’s special about LAMP is that it is a low-cost and simple method which means it can be easily transported to areas which may lack resources, such as developing countries that may also have high incidences of certain pathogens. Scientists have worked on developing methods for detecting a number of human pathogens, with the aim of innovating more effective diagnosis and treatment of related infections and diseases.
Here is an overview of cases where LAMP has shown itself to be an effective method for pathogen identification:
Malaria is responsible for over half a million deaths each year, the World Health Organisation have advised that more accurate diagnosis of malaria infection is an essential part of the plan to strengthen malaria control and ultimately eliminate it. LAMP allows of the distinction between similar species, can uncover reservoirs of asymptomatic infections, detect drug resistance and more.
While mostly eradicated in many parts of the developed world, tuberculosis (TB) still presents a large-scale public health problem. Research has shown that a huge 37% of cases go undiagnosed. LAMP has been developed in order to provide early diagnosis of TB.
Mycobacterium ulcerans are harmful pathogens which cause the debilitating disease of Buruli ulcers in humans. The disease often plagues remote rural areas, making the development of point-of-care tests a priority for healthcare providers. Researchers have been able to show that LAMP can be used within this context, providing a portable and easy to use method to diagnose the disease.
Another group of human pathogens which LAMP has been developed to provide a reliable diagnostic test for are the periodontal anaerobic pathogens. Some of these pathogens have been found to be associated with the development of early-onset periodontitis, necrotising ulcerative gingivitis, and acute periodontitis.
Sleeping sickness, or Trypanosomiasis, is transmitted by tsetse flies infected with the Trypanosoma brucei gambiense pathogen. Researchers have combined LAMP with lateral flow dipstick (LAMP-LFD) methodology in order to create a low-cost process for detecting this pathogen. LAMP-LFD showed a high sensitivity to levels of the pathogen, meaning that in the future medical professionals may be able to utilise LAMP-LFD for accurate diagnosis of sleeping sickness.
LAMP has also been developed for successful detection of the human influenza A virus, as well as two different strains of the avian influenza virus which infects both humans and birds. Scientists have taken the LAMP methodology and added a reverse transcriptase process, RT-LAMP, in order to create a highly sensitive diagnostic tool for detecting the avian affluenza virus.
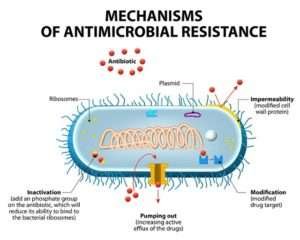 In the world of “superbugs,” there are several LAMP-based diagnostic kits that deliver results in 30 minutes or less. By creating LAMP tests for bacterial genes linked to antibiotic resistance, researchers are able to gain quick insight into the nature of resistance from blood cultures, swabs etc.
In the world of “superbugs,” there are several LAMP-based diagnostic kits that deliver results in 30 minutes or less. By creating LAMP tests for bacterial genes linked to antibiotic resistance, researchers are able to gain quick insight into the nature of resistance from blood cultures, swabs etc.
Keeping Pathogens out of the Human Food Chain
 Further to this, the LAMP method has been developed for use within the food safety industry. Researchers have been able to prove its effective use in detecting salmonella in raw milk, as well as being successful in detecting a range of potentially harmful pathogens in other food sources, for example Campylobacter in chicken farming. Listeria monocytogenes is a specific species of pathogen which can cause listeriosis infection in humans through contaminated food. Again, LAMP has been developed to quickly and easily detect this pathogen, lending itself once more to the the food safety industry.
Further to this, the LAMP method has been developed for use within the food safety industry. Researchers have been able to prove its effective use in detecting salmonella in raw milk, as well as being successful in detecting a range of potentially harmful pathogens in other food sources, for example Campylobacter in chicken farming. Listeria monocytogenes is a specific species of pathogen which can cause listeriosis infection in humans through contaminated food. Again, LAMP has been developed to quickly and easily detect this pathogen, lending itself once more to the the food safety industry.
Future directions
In the future we can expect to see the LAMP process integrated onto Microfluidic Chips, in order to create a diagnostic tool of pathogens. Currently, portable isothermal PCR instruments have made real-time point-of-sample testing using LAMP easy to administer. The Infectious Diseases Society of America have outlined that in order for better care options to be developed, LAMP methods should be focussed on and improved on. They see the benefits of the LAMP process in bringing accessible care to areas hard hit by pathogenic diseases. With these recommendations we can expect to see LAMP playing an increasingly important role in the detection of human pathogens.
Final takeaway
LAMP has been shown to be adaptable in order to provide a method of diagnosis of human pathogens, often providing solutions to areas of the world which are still developing and have the difficulty of a combination of high incidences of pathogen related diseases and a lack of resources to deal with it.



