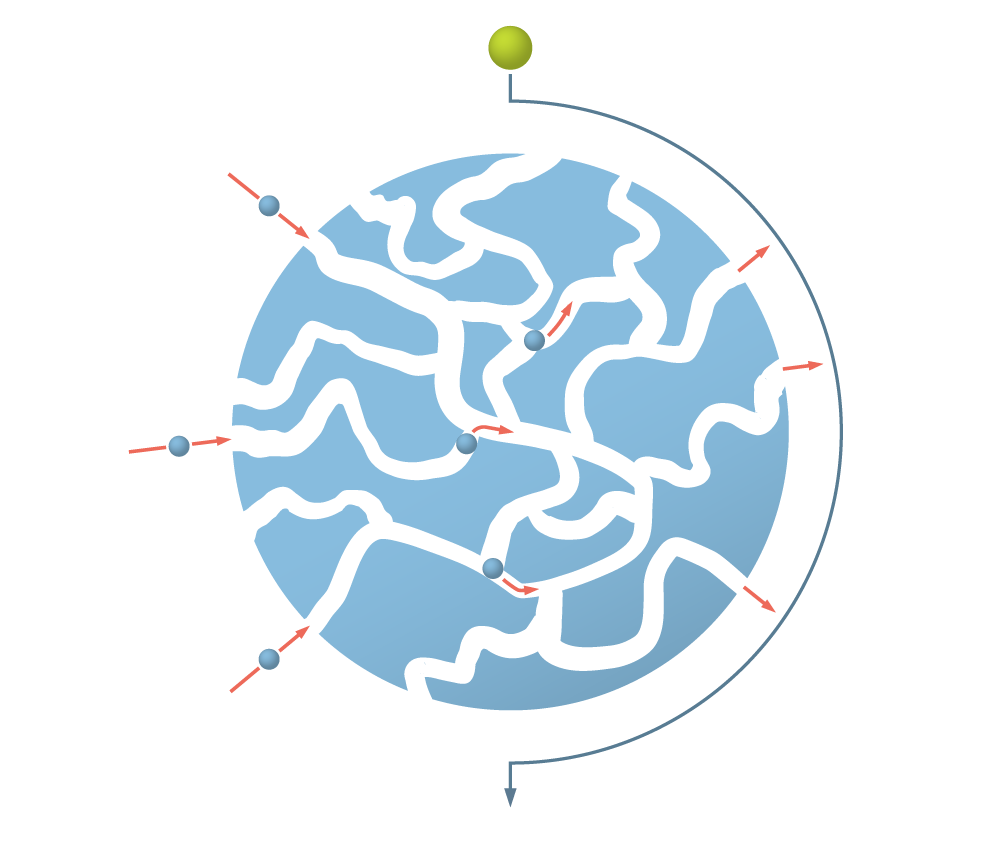
Clarification and Capture – in One-Step
How much time does it currently take you to run your purification? Would you like to increase the speed of purification by 10X? If this sounds unlikely, consider the time effect of removing all clarification steps (filtration, centrifugation etc.) from your downstream processing. What if you could jump straight to product capture, directly from the fermenter?
With the recent development of SMART Chromatography™, downstream processing protocols can now incorporate sample clarification and primary capture into a single step. Fermenter product can be directly loaded from the fermenter onto a uniquely designed column – a SMART Chromatography™ column. Particulates, cell debris etc. pass through the SMART Chromatography column (effectively clarifying the sample) at the same time the target molecule is captured.
The benefits of this are:
- Significant Time Savings
- While time-savings will vary, by combining clarification and capture in a single step, our clients have seen anything from a 5x to 40x time reduction in their time for purification
- Costs associated with clarification are greatly reduced
- e.g., filtration costs can be totally, or very drastically reduced.
- Product yield is increased.
- There is always some product lost during clarification. These product losses are normally “baked in” to many existing downstream processes, but can now be regained.
First, let’s consider the classic bioprocessing workflow that has served so well for decades.
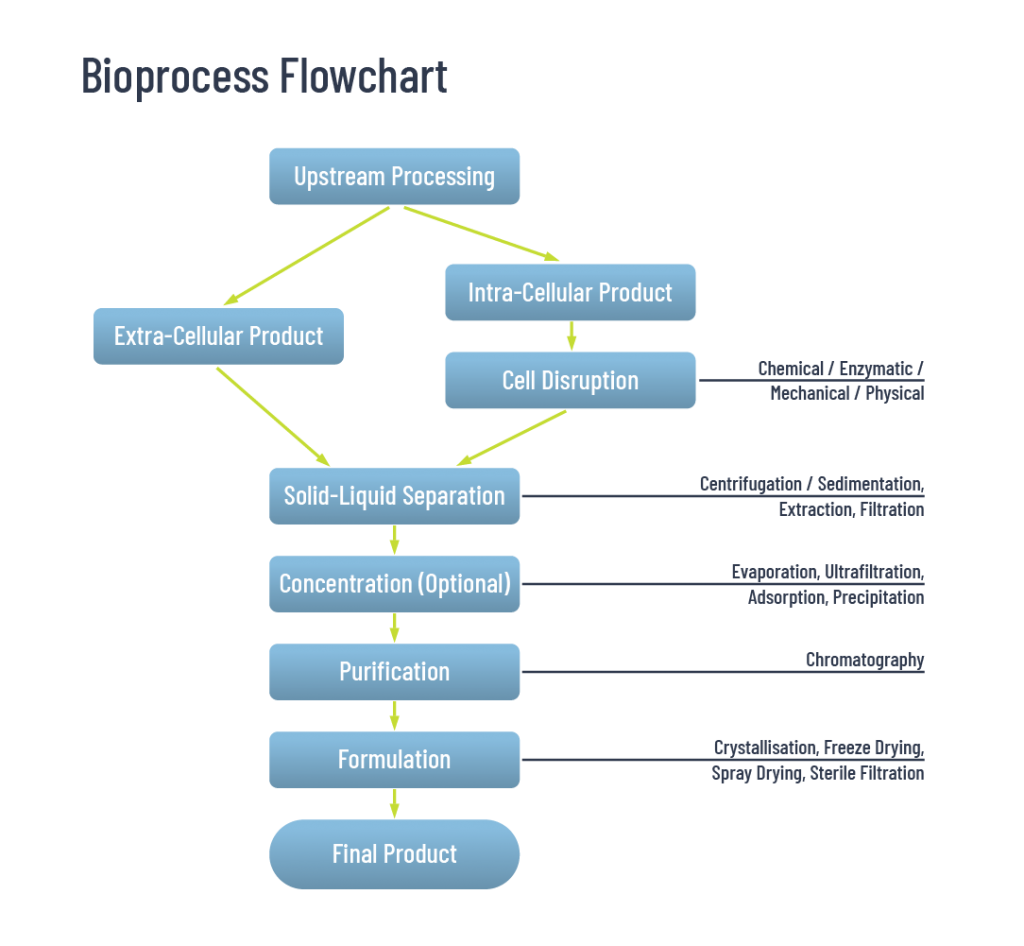
Figure 1: A classical (“textbook”) bioprocessing workflow
Use of a standard chromatography column requires solid-liquid separation (clarification) because such a column would block. A lot of time, effort and ingenuity has been used to develop clarification methods that ensure feedstreams are free of solid matter before being applied to a purification column. The costs, in time and budget, that come with the use of these clarification methods can become a significant part of downstream processing costs.
SMART Chromatography™ addresses the question of “what if solid-liquid separation occurred simultaneously with purification?”
Introducing SMART Chromatography™
- SMART is an acronym of Simplified Method of Applied Radial Technology.
- SMART Chromatography™ technique has wide application and can be used to purify products from mammalian, bacterial, insect and yeast cell cultures.
- The method can be used to purify peptides, antibodies, enzymes and larger biomolecules such as DNA or vira.
The two most notable features of SMART Chromatography™ are:
- Feedstreams containing cells (or crude cell extract) are loaded directly from the fermenter onto the purification column, without prior solid-liquid separation steps.
- SMART Chromatography column designs are linearly scalable. This accelerates the move from R&D to production scale purification.
With the technique of SMART Chromatography™, the downstream processing workflow is changed:
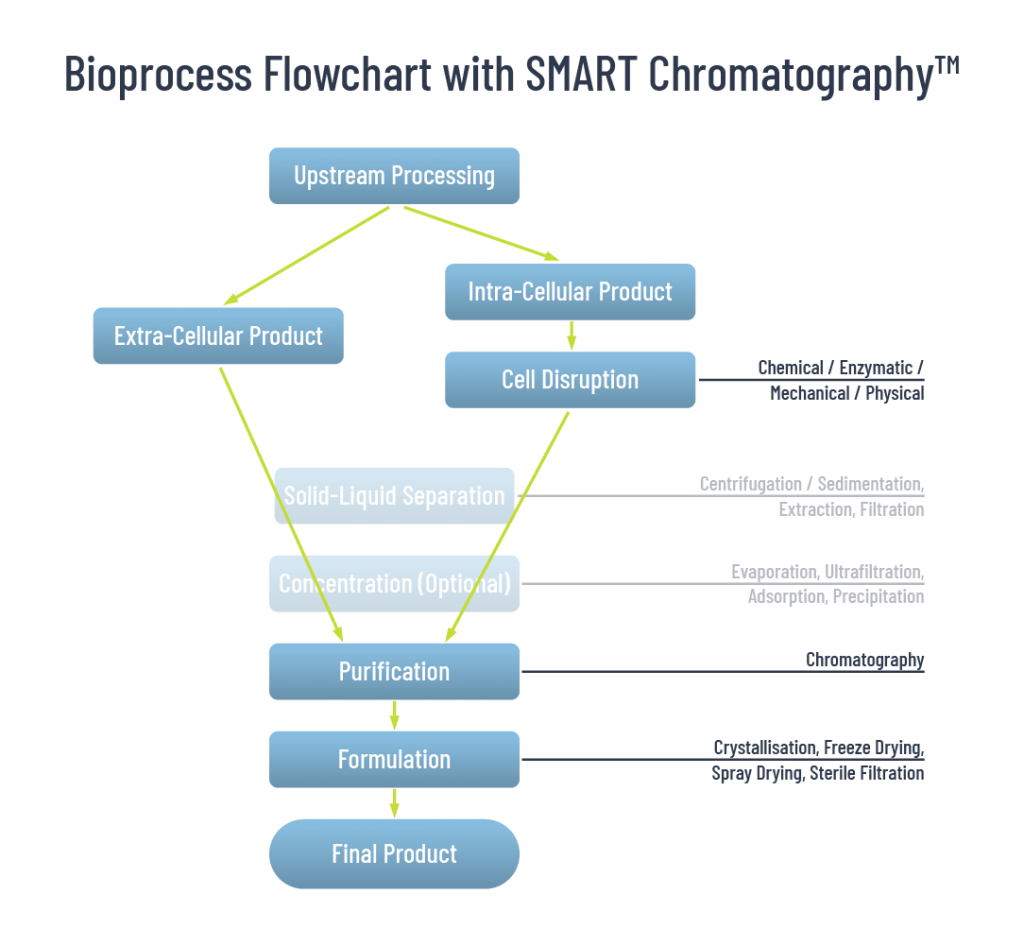
Figure 2: SMART Downstream Processing. The bioprocessing workflow radically changes when SMART Chromatography™ is used and clarification steps are eliminated.
SMART Chromatography™ Eliminates Clarification Prior to Primary Capture Steps
Removal of solid material, i.e., clarification, is normally seen as a required step in downstream processing. However, the clarification of feedstreams is both time-consuming and expensive. As bioreactor volume increases, the problems associated with clarification also increase. Therefore, the elimination of clarification as a necessary step in downstream processing has, without exception, three main benefits:
- Accelerated Production
Time spent on clarification in downstream processing can vary from hours to days, depending upon the volume of fermenter broth, sample type etc. By removing the need for filtration and centrifugation, SMART Chromatography™ allows for faster workflows.
The time savings realized after using SMART Chromatography™ are often considerable. For example, in working with clients we have speed increases in purification times by factors of between 5 to 44 (forty-four). Obviously, such faster paths to production have a considerable effect on planning, delivery and overhead requirements.
2. Increased Profitability
Solid-liquid separation processes often involve multiple steps and methods. For example, it is not unusual to require multiple rounds of filtration, with new filters being required at each round. In such cases, filtration expenses can be considerable.
There are also other costs associated with clarification that cannot be ignored, e.g., consumables, buffer etc. These costs can also be significant, especially in cGMP manufacturing environments.
In short, it is hard to imagine a scenario where removal of clarification from downstream processing does not lead to cost reduction in making the final product, which in turn leads to higher profitability of the entire process.
3. Increased Product Recovery
In our discussions with clients, many are aware of the product loss that occurs during column purification. However, we find that few are aware of how much product is lost between the fermenter and the column before purification begins (i.e., during clarification). In the same way that clarification has been an assumed step in downstream processing, product loss has simply been considered a normal state of affairs, and is factored into product cost, plant design, timelines etc.
We have heard differing estimates of the amount of product that gets lost from the original titre (present in the fermentation broth) to that which gets added to the purification column. Based on this, we believe that between 5% to 60% of product can be lost due to clarification steps.
With SMART Chromatography, product losses can be considerably reduced.
By eliminating clarification, one of the major causes of product loss within bioprocessing is removed. Instead, the total yield of product from the fermenter is now made available for purification. Using SMART Chromatography, some customers have reported up to 98% recovery of the product that was in the fermenter.
Summary of the Main Parameters Affected by Using SMART Chromatography™
| Parameter | Result |
| Process time | Reduced |
| Buffer consumption | Reduced |
| Productivity | Increased |
| Recovery of Product | Increased |
| Retention of biological activity | Increased |
| Overall Effect | Improved process at lower cost |
How does SMART Chromatography™work?
SMART Chromatography™ results from combining two technologies:
1. ZetaCell™ resin
2. Radial flow column chromatography
Together, these technologies enable the elimination of solid-liquid separation from downstream processing.
1. ZetaCell™ Resin
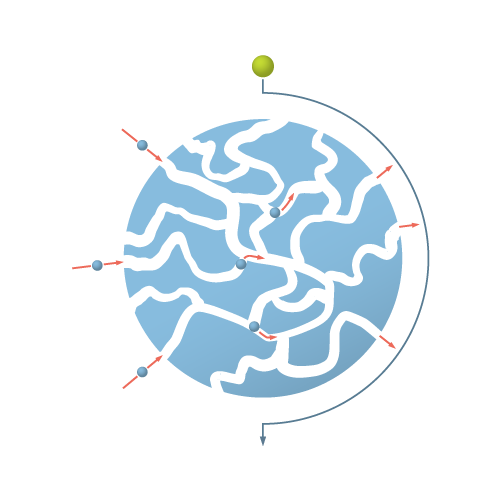
Figure 3: Representation of a ZetaCell™ bead. Each bead is a large particle, based on highly cross-linked agarose. The beads contain channels that allow the product of interest to enter and be captured (blue sphere). Large objects, such as cells or other debris (green sphere) are unable to enter the bead and pass around it.
SMART Chromatography™ uses ZetaCell resin as the solid phase. Each ZetaCell resin is composed of beads that are are based on large particle, highly cross-linked agarose. The desired binding functionality (e.g. protein A, cation, anion etc.) is made on all exposed surfaces – inside and outside – of each ZetaCell bead.
See the table in the Appendix for an overview of the main capture functionalities available for ZetaCell resins.
The way that ZetaCell beads work is simple. Large molecules, such as cells, cell debris and other solid matter in the feedstream are too large to enter the bead. Thus, they pass around each bead. However the target molecule of interest gets bound to the bead by the binding functionality being used.
Large beads alone are not the answer though. Loading cell-containing media to any regular column would be an expensive disaster, no matter what resin was used. So, why doesn’t the SMART Chromatography™ column get blocked? This is where the combination with radial flow chromatography comes in.
2. Radial Flow Chromatography
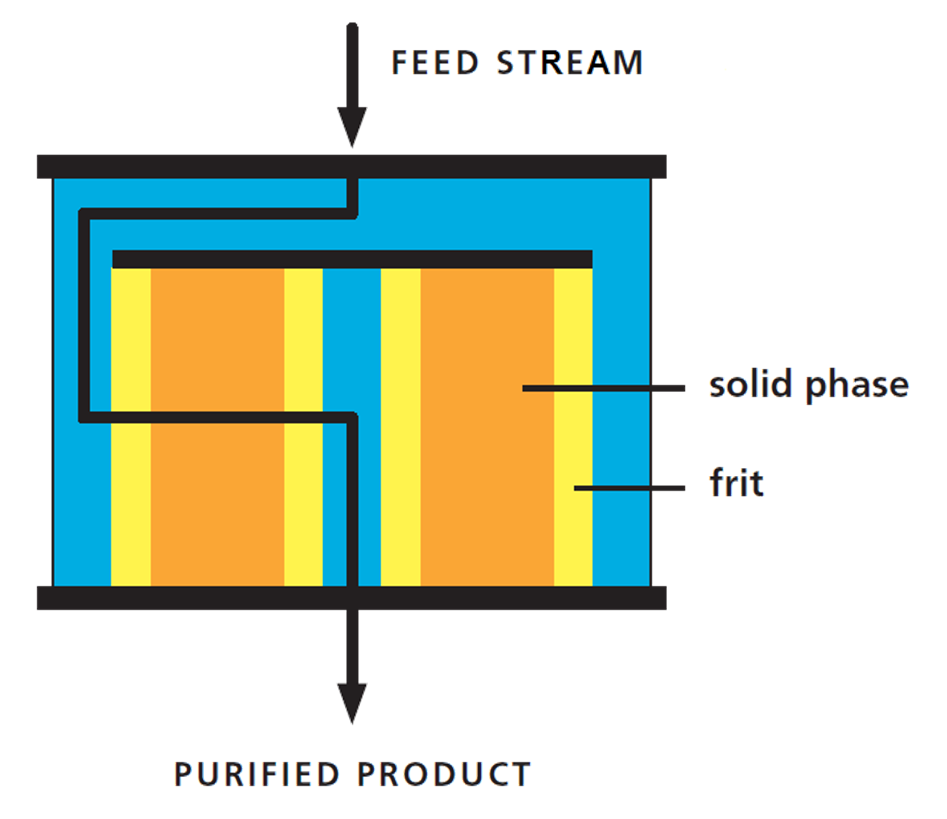
Figure 5: 2-D representation of Radial Flow Chromatography (RFC). Unlike chromatography columns that typically use vertical (top-bottom) flow, RFC columns use horizontal flow. Bed height and chromatographic properties remain, while footprint and weight are dramatically reduced.
Radial Flow Chromatography (RFC) is an efficient, low pressure technology for bio-molecule purification. Thanks to the column geometry, RFC is very suitable for high throughput separations in process, pilot or lab scale.
There are many advantages to using RFC, including dramatic reductions in column weight/ footprint and easy packing properties. However, one of the most useful features within a bioprocessing setting is that RFC is linearly scalable. The advantages of linear scalability are very useful in a production setting and are expanded upon in a later section.
ZetaCell Resin + Radial Flow = SMART Chromatography™
SMART downstream processing is enabled when radial flow columns are packed with ZetaCell resin. Radial flow columns have a larger surface area for column loading than the equivalent “regular” (i.e. axial flow) chromatography columns. For example, a radial flow column will typically have twenty times the frit surface area compared to an axial flow column with the same bed length and bed volume. This increased surface area for loading means that feedstreams containing cells and other debris are spread over a much larger surface area when entering the column, resulting in lower pressure.
Once inside the column, cells/debris passes around the ZetaCell™ Resin and the target of interest is captured. Cells pass through and out of the column – again at low pressure over a large surface area – without blocking. After column washing, product is eluted from the column.
SMART Chromatography™ Allows for Rapid R&D, Troubleshooting and Optimization of Bioprocessing
A critical challenge in the development of any bio-manufacturing process is moving from R&D to production – the process commonly referred to as “scale-up.” SMART Chromatography™ uses a major feature of RFC – linear scalability – to make the process of scale-up more straightforward.
Small- and medium-scale RFC columns have properties that are representative of larger, process-scale columns. For any given bed length, properties such as flow rate, cell density, resin binding, column washing parameters etc. remain the same over all column sizes. This linear scalability allows for accelerated process development, because many fundamental properties of the purification process that are set at the R&D stage can be implemented at larger scales.
The linear scalability of RFC works also works in reverse. Scale-down is more straightforward with SMART Chromatography™ compared to regular column chromatography. This is an excellent property for trouble-shooting, new process development etc.
SMART Chromatography™ Works on a Wide Range of Cell Types and Target Molecules
SMART Chromatography™ has been implemented at, or is currently undergoing evaluation at 20+ different sites globally. EMP Biotech has accumulated experience in testing and roll-out of the use of SMART Chromatography™ for:
- Yeast (Saccharomyces cerevisiae)
- Purification of recombinant protein by IEX, using ZetaCell SP resin.
- Currently at 90% recovery.

Figure 8: Scale Up SMART Chromatography™ Study on Yeast. Using a 200 mL column, 6 cm bed length (centre of picture, with yellow “post-it” attached). Yeast cells loaded directly from fermentation broth (left hand side) and passing through column to waste (beaker, right hand side). Photo credit: emp Biotech GmbH.
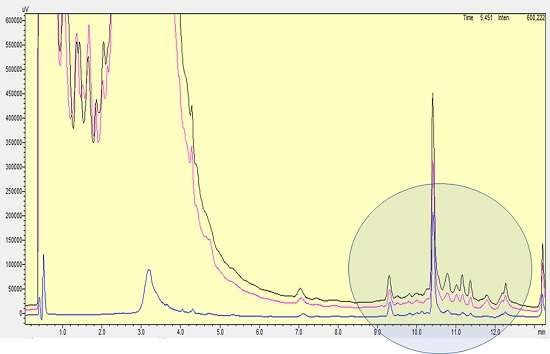
Figure 9: Scale Up SMART Chromatography™ Validation on Yeast. Analytical RP-HPLC shows excellent purification (blue trace) compared with untreated and adjusted fermentation broth (black and pink traces)
- Mammalian cells (CHO).
- Purification of recombinant monoclonal IgG using ZetaCell Protein A resin.
- Recovery of IgG: 98%

Figure 10: R&D scale SMART Chromatography™ column being loaded with CHO cells. The red colour is due to the presence of phenol red in the cell culture medium (photo credit: emp Biotech).
At the BioProcessing Network (BPN) conference (Adelaide, Australia, 2017), CSIRO (the premier Australian research centre) presented data for the use of SMART Chromatography™ to purify monoclonal antibodies from mouse hybridoma cell lines (Martin, J et. al, 2017). The SMART Chromatography™ column showed about 93% antibody capture, without the need for removal of cells from the media applied to the purification column.
SMART Chromatography™ has also been used with other cell systems such as E. coli and HEK-293, as well as cell systems which cannot be disclosed in this article due to confidentiality agreements between emp Biotech and their clients.
How Can I Fit SMART Chromatography into My Existing Workflow?
Resistance to change is high in any field and something we come across regularly in trying to implement SMART Chromatography™, despite its advantages. Obviously, it’s easier to implement SMART Chromatography™ for a new process. However, what if you want to change a current process, for reasons of improved time, economics, or increased product yield?
The best approach is to load the SMART Chromatography column “offline,” by pumping fermenter product directly onto the column. This can be done with any standard pump. It’s good to know that SMART Chromatography™ columns have dramatically reduced footprint and weight, compared to a vertical column of equivalent bed height and thus use less physical space. This is clearly an advantage in a bioprocessing environment.
For example, a 250 litre column SMART Chromatography™ set up will have a diameter of 70 cm, i.e, the column will fit through a standard doorway. As a “rule of thumb” the columns have around 25% of the footprint of the equivalent axial flow column.
So columns can be brought to the same vicinity as the fermenter (if necessary). Once fully loaded, the columns will be washed by to remove solid material (cells, debris, etc.). Elution of product can be implemented as in the existing process.
In other words, SMART Chromatography™ can be implemented into existing workflows with relatively minor modifications, compared to the upside gained by implementing the method.
Customer Applications of SMART Chromatography™
The following table shows some examples where SMART Chromatography™ is being used by clients in bioprocessing.
| Cell system | Target molecule | Method | ZetaCell resin | Binding capacity |
| CHO | Antibody (human IgG) | Affinity | Protein A & Protein A Endure | 15 mg/mL |
| CHO | Antibody (mouse IgG1) | Affinity | Protein A | 16 mg/mL |
| Pichia pastoris | Confidential | Affinity (IMAC) | IDA | Proprietary |
| E. coli | His-tagged GFP | Affinity (IMAC) | Ni-NTA | 16.7 mg/mL |
| E. coli | His-tagged GFP | Affinity (IMAC) | IDA | 15.9 mg/mL |
| Saccharomyces cerevisiae | Confidential | IEX | SP | >25 mg/mL |
| E. coli | Smallpox virus (300 nm) | IEX | Q Boost | Proprietary |
How Do I Know if SMART Chromatography will work for Me?
The best way to get started with SMART Chromatography™ is to test the binding of your target molecule to the ZetaCell resin of choice, using a chromatography workstation. For this, we can supply FPLC columns packed with ZetaCell, to be used on clarified resin.
Note: the purpose of this excercise is to simply determine which ZetaCell resin is the most suitable for your target molecule. This is why the test is done using clarified resin.
After determining the best resin for your purpose, we can provide a small (5-10 mL) SMART Chromatography™ column, where you can load your cell culture/extract directly onto the column.
We will, of course, provide full consultation and guidance through this process.
Interested?
Contact us if you want to learn more. Please submit your enquiries using the form at the foot of this page, or send a mail to info@youdobio.com. Alternatively, just reach for your phone and call or text using our number at the top of this page.
An expert in SMART Chromatography™ will visit you at your convenience.
Frequently Asked Questions (FAQs)
Below is a list of common questions that arise in discussions about SMART Chromatography™. If you have your own questions and cannot find an answer below, or would like more specifics, please submit your question using the form at the foot of the page.
Q1. How quickly can I get started with SMART Chromatography™?
For a new process, we expect it to take about 6 months to go from R&D to production. We have experience of re-developing an existing process, for purification of a registered pharmaceutical – this took 12 months. These figures represent a rough estimate – it will depend on the process.
Q2. Is SMART Chromatography™ more expensive to implement than what I’m currently using?
Small volume columns, e.g. 5 mL – 200 mL, are slightly more expensive than traditional axial flow columns. At large scale (> 5L) the price of columns and resin is similar to similar-scale axial flow columns. In any case, SMART Chromatography™ will save you money in the long run.
Q3. How easy is SMART Chromatography™ to implement?
Not difficult. A typical process work-up will involve using R&D columns at 5 mL scale, validation columns at 200 mL scale, before going to process scale. EMP Biotech has developed the SMART Chromatography™ method and will be on-hand to ensure that implementation goes smoothly.
EMP Biotech can provide small-scale columns and resin for proof-of-principle studies. Such columns can be fitted to standard chromatography equipment, e.g. ÄKTA purification systems. SMART Chromatography columns at small-scale can be loaded with a peristaltic pump.
Q4. Can we pack and repack SMART™ Chromatography columns ourselves?
SMART Chromatography™ columns are remarkably straightforward to pack. Columns can be packed using a packing station. Alternatively, the nature of the column design means that columns can be packed in a “low tech” way with resin slurry using a pump and a pressure.
Q5. How are SMART Chromatography™ columns cleaned?
SMART Chromatography™ columns are cleaned in the same way as traditional axial flow columns. Cell and cellular debris are washed through the column and a short back flush will remove any solids trapped in the inlet frit. The resins used for SMART Chromatography™ are generally tolerant of harsh CIP reagents such as 0.5 – 1 M NaOH, although obviously this is ligand dependent.
Q6. Can SMART Chromatography™ be used in a GMP environment?
Yes. There are currently two clients using SMART Chromatography™ in a GMP setting.
Q7. Who are EMP Biotech?
EMP Biotech was founded in 1983 and is a privately-held company, based in Berlin, Germany. The company has four active manufacturing sites. EMP is ISO certified (ISO 9001:2015). The company has passed dozens of audits by major organizations using a wide range of manufacturing products.
The EMP team consists of accomplished chemists and engineers who professionally deliver a wide range of products within purification, chemistry and biochemistry.
You can learn more about EMP Biotech from their website.
EMP Biotech is represented in Denmark, Sweden, Norway and Iceland by You Do Bio.
Appendix: main purification functionalities available for ZetaCell™ resins
| Resin name | Functionality | Ligand | Target |
| ZetaCell Protein A | Affinity | Protein A | Antibodies |
| ZetaCell Protein A Endure | Affinity | Base-stable protein A | Antibodies |
| ZetaCell Protein G | Affinity | Protein G | Antibodies |
| ZetaCell IDA | Affinity | Iminodiacetic acid | His-tagged proteins |
| ZetaCell NTA (uncharged) | Affinity | Nitrilotriacetic acid | His-tagged proteins |
| ZetaCell Cu-NTA | Affinity | Nitrilotriacetic acid Cu2+ | His-tagged proteins |
| ZetaCell Co-NTA | Affinity | Nitrilotriacetic acid Co2+ | His-tagged proteins |
| ZetaCell Ni-NTA | Affinity | Nitrilotriacetic acid Ni2+ | His-tagged proteins |
| ZetaCell Zn-NTA | Affinity | Nitrilotriacetic acid Zn2+ | His-tagged proteins |
| ZetaCell Phenyl | HIC | Phenyl (C6H5) | Protein purification |
| ZetaCell Q | IEX (Strong Anion) | Quaternary ammonium | Neg. charged molecules |
| ZetaCell Q Boost | IEX (Strong Anion) | Quaternary ammonium | Neg. charged molecules |
| ZetaCell SP | IEX (Strong Cation) | Sulphopropyl | Pos. charged molecules |
| ZetaCell SP Boost | IEX (Strong Cation) | Sulphopropyl | Pos. charged molecules |
| ZetaCell DEAE | IEX (Weak Anion) | Diethyl-aminoethyl | Neg. charged molecules |
| ZetaCell DEAE Boost | IEX (Weak Anion) | Diethyl-aminoethyl | Neg. charged molecules |
| ZetaCell CM | IEX (Weak Cation) | Carboxy-methyl | Pos. charged molecules |
| ZetaCell CM Boost | IEX (Weak Cation) | Carboxy-methyl | Pos. charged molecules |
| ZetaCell Aldehyde activated | Primary amine binding | Client provided | Binding to specific ligand |