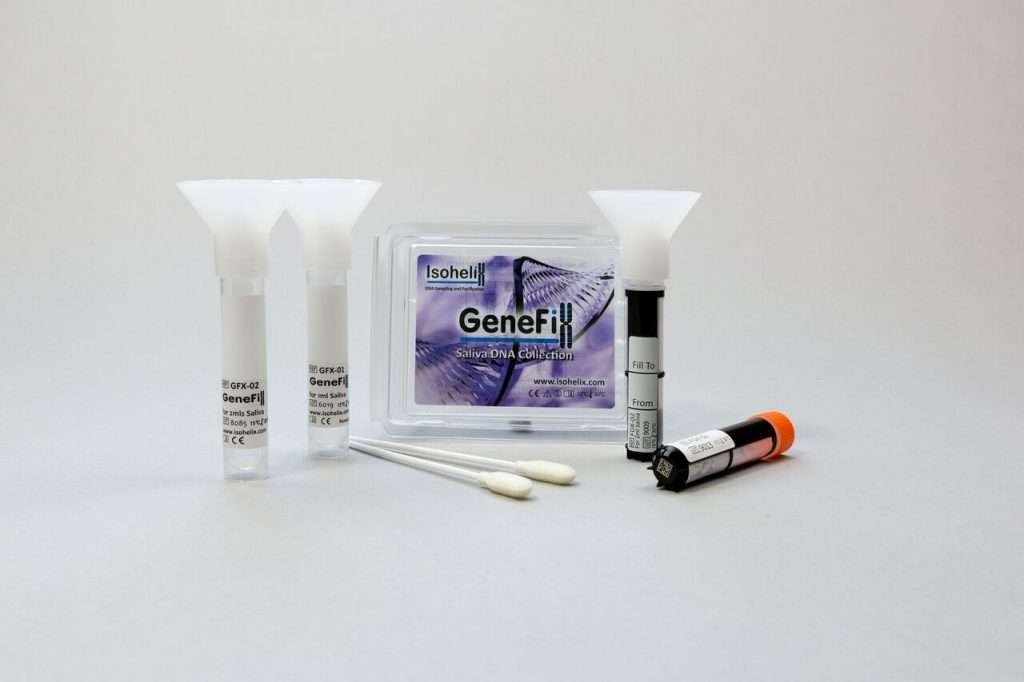
Isohelix have released a new product in their GeneFix™ range of DNA saliva collector. The new product is designed to help in two cases:
- Low yield donors, when 1- or 2 mL of saliva does not yield enough DNA.
- Applications where you want to maximise DNA yield.
To address these issues, Isohelix have designed the Xtra DNA Collect for 3ml saliva samples.
The Xtra DNA Collect saliva collectors are pre-filled with 1ml of Xtra concentrated buffer, meaning the final sample volume remains 4ml. The advantage here is that customers will process the same volume of liquid through GSPN or other DNA isolation kits, but the sample contains 50% more saliva, so the final elution will be more concentrated. We have established that the buffer gives 2 years minimum stabilisation time, but we are continuing to test, so we are confident that the stabilisation time is longer, and will update as testing progresses.
This could help with re-sampling of low yield donors or may be attractive to end users wanting as much DNA as possible.
The ‘Xtra DNA Collect’ is designed to collect 3ml of Saliva, thus improving yields over the standard GeneFix and 1ml and 2 ml collectors. It is packaged in the same tube yet filled with 1ml of a higher concentration buffer to allow for extra dilution of the saliva. It has most of the benefits of our standard collector and will appeal to scientists requiring maximal yields even in cases where patient sampling times may be extended. Currently in house and ongoing studies show full 2 year stabilization but we feel confident this will be extended over time.
If you still prefer swabs for DNA collection, you should also check out the recently-released RapiDri swab kits.
Product codes will be GFX-03/02, GFX-03/12, GFX-03/50 GeneFiX™ 3ml Xtra DNA Saliva Collector for 2 samples, 12 samples and 50 samples respectively.