The demand for COVID-19 testing is unprecedented and will remain so for some time. In Denmark, for instance, by December 17, 2020, over seven million tests had been performed for coronavirus, around 124,000 of which tested positive.1 In the United States, over 260 million COVID-19 tests have been done so far.2 Globally, it is expected that billions of tests will be needed over the coming months to years, so any factors that makes COVID-19 testing simpler or safer has tremendous results for the implementation of public health measures and cost savings.
Most laboratory-based COVID-19 testing is a combination of real‐time quantitative reverse transcription PCR (rRT‐PCR) used on patient samples acquired through nasopharyngeal swabs. But other types of samples can be used for testing, including oropharyngeal (throat) swabs, nose swabs, bronchoalveolar lavage fluid, and saliva. Similarly, while RT‐PCR has become the reference standard molecular test to detect SARS‐CoV‐2 genetic material, other diagnostic methods available are are just as good (e.g. Isothermal PCR).
For non-PCR based testing, viral antigens can be detected as well as the antibodies developed against them. These are the basis of an increasing number of point-of-care (POC) tests. However, as of yet, the World Health Organisation (WHO) prefers the use of antigen-based tests in research settings only, and not for clinical purposes.3 This is because rapid diagnostic tests based on antigen detection have sensitivity in the range of 34% to 80%, which means they can potentially miss more than half of COVID-19 infected patients.4 The issue with tests based on the presence of antibodies – the immunoglobulins (Ig) M, G, and A – is that antibodies are produced days to weeks after infection. Any actions to alter the spread of COVID-19 need to take place at onset of infection. Furthermore, antibody response varies between individuals. For example, some infected people may show a delayed antibody response, or even none at all.
Thus, in the majority of cases, COVID-19 testing depends on the logistics of collecting a sample from the patient and sending it to the laboratory for a PCR-based test. This brings our discussion back to what is a suitable sample to acquire for testing – in terms of reliability, safety and convenience. Given that saliva appears to be the most simple and safe sample to acquire, for reasons discussed below, the key question concerns the suitability of saliva as a test material compared to swabs.
Key question: how does saliva sampling compare to swabs? Results from Research
Considering the convenience and safety that comes with using saliva, a number of researchers have looked into its feasibility as a testing specimen and also how it compares to the gold standard nasopharyngeal swab when it comes to yielding results.
Guillaume Butler-Laporte, MD, and his colleagues addressed this question head-on by carrying out a study to directly compare saliva and nasopharyngeal swab testing for the detection of SARS-CoV-2 using PCR. This study is noteworthy is because its design – a systematic review and meta-analysis – produces high quality evidence supporting the use of saliva. The comprehensive study, includes in its quantitative synthesis 5922 unique patients from 16 studies, from an initial 385 references.5 The findings are available on the JAMA Internal Medicine website.
So what does this analysis show? It reveals that the pooled sensitivity of saliva was 83.2% and its pooled specificity was 99.2%. In comparison, the sensitivity of nasopharyngeal swabs was 84.8% and specificity, 98.9%. The authors conclude that saliva diagnostic accuracy is comparable to that of nasopharyngeal swabs, especially in the ambulatory setting. They point out that these findings make the case for larger-scale studies on the use of saliva as an alternative to nasopharyngeal swabs.
Several other studies corroborate these findings. In another recent study, researchers at the Memorial Sloan Kettering Cancer Center, New York, compared the use of saliva with swab collection in symptomatic and asymptomatic health care workers for the detection of SARS-CoV-2 RNA by RT-PCR. They found that the agreement between saliva and nasopharyngeal swabs was 97.7%, with a sensitivity of 94.1%. They observed that these results indicate that saliva is – at the very least – an acceptable alternative to nasopharyngeal swabs for SARS-CoV-2 RNA detection, and it has the added benefit of being broadly deployable.6
Similarly, a more recent study conducted by ARUP Laboratories and University of Utah (U of U) Health in 354 patients prospectively compared health care worker-collected nasopharyngeal swabs to self-collected saliva specimens for the diagnosis of COVID-19. The positive agreement between nasopharyngeal swabs and saliva was 93.8%, and the negative agreement was 97.8%.7 In another instance, Anne Wyllie, PhD, and colleagues reported in a letter to the The New England Journal of Medicine that they detected more SARS-CoV-2 RNA copies in saliva specimens than nasopharyngeal swab specimens (mean log copies per milliliter 5.58 vs. 4.93, respectively). Furthermore, a higher percentage of saliva samples were positive up to 10 days after the initial diagnosis of COVID-19 compared with nasopharyngeal swab samples. The authors further determined that saliva and nasopharyngeal swab specimens had similar sensitivity in the detection of SARS-CoV-2 during hospitalisation.8
In short, there is an increasing number of studies indicating that not only is saliva sampling a viable option for COVID-19, but in fact it is on par with nasopharyngeal swabs. But why all this interest in and emphasis on saliva collection over other specimens? As mentioned earlier, it represents convenience and safety. Let’s look into the advantages associated with saliva sampling in more detail.
Using Saliva in COVID-19 Testing Gives Clear Advantages
Saliva collection for COVID-19 testing gives clear advantages, particularly when compared to nasopharyngeal swab testing:
Ease of testing – saliva can be self-collected at home and mailed to lab
Saliva collection is a noninvasive and simple procedure that does not cause any pain or discomfort and can be performed by patients in the comfort of home. It just takes a few minutes and results are received within days. Unlike a nasopharyngeal swab, it does not require the presence of a trained healthcare professional to collect it.9
Because saliva is easy to collect, patients are more likely to provide samples. This becomes important, given the likelihood that the public will get “testing fatigue” as the pandemic progresses. Valentine-Graves et al assessed the acceptability of the collection, packing and shipping process among participants who self-collected saliva for COVID-19 testing. They observed high acceptability (>84%) and noted that self-collection and shipping of specimens for SARS-CoV-2 testing was acceptable in a diverse group of participants.10
A further consideration is that not all patients have the means to transport themselves to a testing facility. Using saliva, elderly, infirm, or disabled patients will more easily be able to provide test samples.
Eloise Williams and colleagues investigated the transport and storage feasibility of saliva in the context of SARS-CoV-2 testing. They were able to detect the virus in saliva even on day 7 on samples that were stored either at 4°C or just room temperature.11
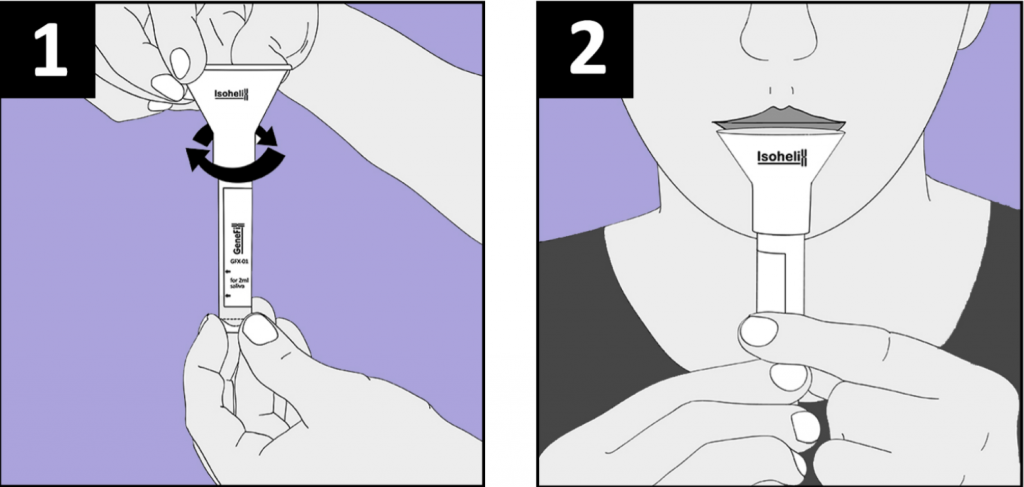
Everyone in the COVID-19 Testing Chain Gets Reduced Exposure
Self-collection at home eliminates the risk of exposure to healthcare workers as they do not have to see the patient to acquire a sample, in contrast to a nasopharyngeal swab. And, it works the other way round as well: patients do not have to go to health facilities to give a sample where they would be exposed to other patients or silent carriers of COVID-19.12
Reducing the exposure of health workers to infected patients is now of greater importance than ever, with the development of more infections strains of the virus.
The ease and safety of saliva collection makes it ideal for mass testing. For instance, the University of Illinois Urbana-Champaign announced it would be processing 20,000 saliva tests per day to keep a close eye on the unfolding COVID situation. Each student and staff member would be tested twice a week. They used a phone app to coordinate and run the whole project.13
Using Saliva results in Significant Cost-Savings
Self-collection of saliva is a very low-cost sampling method. There are no travel costs, no queues and delays at testing centers. It decreases the burden on healthcare staff. The drastic reduction in exposure reduces the spread of the virus. Collectively, these changes lead to a smaller toll on healthcare resources.
Just How Much Cost-Saving Can the Use of Saliva Provide in COVID-19 Testing?
Wong et al, in their study comparing saliva and nasopharyngeal swab specimens for COVID-19 testing, calculated that the equipment costs for saliva collection were $8.24 per 100 samples vs. $104.87 per 100 nasopharyngeal swab specimens.14 When we extrapolate this to the billions of samples that are expected to be collected around the world over the coming time, it is easy to see the huge financial impact that saliva collection can make over nasopharyngeal swabs in COVID testing and surveillance.
Now that we clearly acknowledge the suitability of saliva as a specimen for COVID-19 testing, you don’t have to go too far to find the best saliva collection kits. We have them right here for you. These kits by GeneFiX™ are designed to be exceptionally easy to use and they stabilise nucleic acids for the long term. If you have any questions, please do not hesitate to get in touch.
REFERENCES
- Statista. Weekly number of people tested for coronavirus (COVID-19) in Denmark. https://www.statista.com/statistics/1106073/weekly-number-of-coronavirus-tests-in-denmark/. Accessed Saturday, 16 January, 2021.
- Our World in Data. Total COVID-19 tests. https://ourworldindata.org/grapher/full-list-total-tests-for-covid-19?time=2020-02-20..latest. Accessed Saturday, 16 January, 2021.
- World Health Organization. Advice on the use of point-of-care immunodiagnostic tests for COVID-19. 8 April 2020. https://www.who.int/news-room/commentaries/detail/advice-on-the-use-of-point-of-care-immunodiagnostic-tests-for-covid-19. Accessed Saturday, 16 January, 2021.
- Bruning AHL, Leeflang MMG, Vos JMBW, Spijker R, de Jong MD, Wolthers KC, Pajkrt D. Rapid Tests for Influenza, Respiratory Syncytial Virus, and Other Respiratory Viruses: A Systematic Review and Meta-analysis. Clin Infect Dis. 2017 Sep 15;65(6):1026-1032. doi: 10.1093/cid/cix461.
- Butler-Laporte G, Lawandi A, Schiller I, et al. Comparison of Saliva and Nasopharyngeal Swab Nucleic Acid Amplification Testing for Detection of SARS-CoV-2: A Systematic Review and Meta-analysis. JAMA Intern Med. Published online January 15, 2021. doi:10.1001/jamainternmed.2020.8876.
- Babady NE, McMillen T, Jani K, Viale A, Robilotti EV, Aslam A, Diver M, Sokoli D, Mason G, Shah MK, Korenstein D, Kamboj M. Performance of Severe Acute Respiratory Syndrome Coronavirus 2 Real-Time RT-PCR Tests on Oral Rinses and Saliva Samples. J Mol Diagn. 2021 Jan;23(1):3-9. doi: 10.1016/j.jmoldx.2020.10.018. Epub 2020 Nov 17.
- K. E. Hanson, A. P. Barker, D. R. Hillyard, N Gilmore, J. W. Barrett, R. R. Orlandi, S. M. Shakir. Self-Collected Anterior Nasal and Saliva Specimens versus Healthcare Worker-Collected Nasopharyngeal Swabs for the Molecular Detection of SARS-CoV-2. Journal of Clinical Microbiology, 2020; DOI: 10.1128/JCM.01824-20.
- Wyllie AL, Fournier J, Casanovas-Massana A, et al. Saliva or nasopharyngeal swab specimens for detection of SARS-CoV-2. N Engl J Med 2020;383:1283-1286.
- Fernandes LL, Pacheco VB, Borges L, Athwal HK, de Paula Eduardo F, Bezinelli L, Correa L, Jimenez M, Dame-Teixeira N, Lombaert IMA, Heller D. Saliva in the Diagnosis of COVID-19: A Review and New Research Directions. J Dent Res. 2020 Dec;99(13):1435-1443. doi: 10.1177/0022034520960070. Epub 2020 Sep 16.
- Valentine-Graves M, Hall E, Guest JL, Adam E, Valencia R, Shinn K, et al. (2020) At-home self-collection of saliva, oropharyngeal swabs and dried blood spots for SARS-CoV-2 diagnosis and serology: Post-collection acceptability of specimen collection process and patient confidence in specimens. PLoS ONE 15(8): e0236775. https://doi.org/10.1371/journal.pone.0236775.
- Williams E, Isles N, Chong B, Bond K, Yoga Y, Druce J, Catton M, Ballard SA, Howden BP, Williamson DA. Detection of SARS-CoV-2 in saliva: implications for specimen transport and storage. J Med Microbiol. 2020 Dec 3. doi: 10.1099/jmm.0.001285. Epub ahead of print.
- Michailidou E, Poulopoulos A, Tzimagiorgis G. Salivary diagnostics of the novel coronavirus SARS-CoV-2 (COVID-19). Oral Dis. 2020 Nov 19. doi: 10.1111/odi.13729. Epub ahead of print.
- The Scientist. U of Illinois Returns to School with 20,000 Saliva Tests Per Day. https://www.the-scientist.com/news-opinion/u-of-illinois-returns-to-school-with-20-000-saliva-tests-per-day-67890. Accessed Saturday, 16 January, 2021.
- Wong SCY, Tse H, Siu HK, et al. Posterior Oropharyngeal Saliva for the Detection of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Clin Infect Dis. 2020;71(11):2939-2946. doi:10.1093/cid/ciaa797



